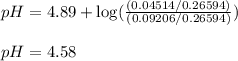Chemistry, 28.03.2020 04:02 cupcake20019peehui
An analytical chemist is titrating of a solution of propionic acid with a solution of 224.9 ml of a 0.6100M solution of propionic acid (HC2H5CO2) with a 1.1000M solution of KOH. The pKa of proionic acid 4.89.
Calculate the pH of the acid solution after the chemist has added 41.04mL of the KOH solution to it.
Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of solution added. Round your answer to decimal places.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:50
Determine the empirical formula for succinic acid that is composed of 40.60% carbon, 5.18% hydrogen, and 54.22% oxygen.
Answers: 1

Chemistry, 22.06.2019 12:50
The number at the end of an isotope’s name is the number.
Answers: 1

Chemistry, 22.06.2019 19:30
Astudent conducts an experiment to determine how the amount of water given to a plant affects its growth. what is the independent variable for this experiment?
Answers: 1

Chemistry, 22.06.2019 23:00
In the reaction h2co3 (aq) + 3nh3 (aq) = 2 nh4+ (aq) + co3 2-, how many electrons are transferred?
Answers: 3
You know the right answer?
An analytical chemist is titrating of a solution of propionic acid with a solution of 224.9 ml of a...
Questions


Mathematics, 04.02.2022 18:10


Biology, 04.02.2022 18:10

Physics, 04.02.2022 18:10


Mathematics, 04.02.2022 18:10




Mathematics, 04.02.2022 18:20



Mathematics, 04.02.2022 18:20

English, 04.02.2022 18:20

History, 04.02.2022 18:20



Mathematics, 04.02.2022 18:20

Mathematics, 04.02.2022 18:20

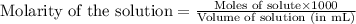 .....(1)
.....(1)
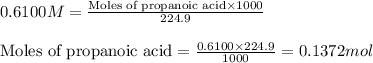
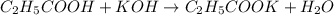
![pH=pK_a+\log(\frac{[\text{salt}]}{[acid]})](/tpl/images/0568/6689/3d096.png)
![pH=pK_a+\log(\frac{[C_2H_5COOK]}{[C_2H_5COOH]})](/tpl/images/0568/6689/3ede9.png)
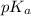 = negative logarithm of acid dissociation constant of propanoic acid = 4.89
= negative logarithm of acid dissociation constant of propanoic acid = 4.89![[C_2H_5COOK]=\frac{0.04514}{0.26594}](/tpl/images/0568/6689/11196.png)
![[C_2H_5COOH]=\frac{0.09206}{0.26594}](/tpl/images/0568/6689/86178.png)