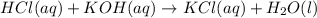During a laboratory activity, a student places 25.0 mL of HCl(aq) of unknown concentration into a flask. The student adds four drops of phenolphthalein to the solution in the flask. The solution is titrated with 0.150 M KOH(aq) until the solution appears faint pink. The volume of KOH(aq) added is 18.5 mL. Complete the equation for the neutralization reaction that occurs during the titration: KOH (aq) + HCl (aq) -->

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 17:40
What are the resulting coefficients when you balance the chemical equation for the combustion of ethane, c2h6? in this reaction, ethane is burned in the presence of oxygen (o2) to form carbon dioxide (co2) and water (h2o). (g)+(g)→(g)+(g)
Answers: 1

Chemistry, 22.06.2019 00:40
During which time interval does the object travel approximately 10 meters
Answers: 3

Chemistry, 22.06.2019 03:00
About 70 percent of the earth's surface is water-covered, and about 96.5 percent of all earth's water is salt water. identify the watery feature on earth that is made of freshwater rather than salt water. a) bay b) glacier c) ocean d) sea it is not incomplete this is the true question
Answers: 1

Chemistry, 23.06.2019 00:30
Ok, so i have 2 questions. try to answer them both: (the topic is fire) 1) how can you represent the chemical reaction of fire? 2) what kind of bond is formed in this chemical reaction
Answers: 3
You know the right answer?
During a laboratory activity, a student places 25.0 mL of HCl(aq) of unknown concentration into a fl...
Questions

Mathematics, 25.08.2019 12:30



Mathematics, 25.08.2019 12:30







History, 25.08.2019 12:30

Mathematics, 25.08.2019 12:30


Physics, 25.08.2019 12:30



English, 25.08.2019 12:30

Mathematics, 25.08.2019 12:30


Mathematics, 25.08.2019 12:30