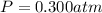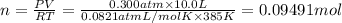Chemistry, 21.03.2020 02:59 carleygalloway103
0.100 mol of CaCO3 and 0.100 mol CaO are placed in an 10.0 L evacuated container and heated to 385 K. When equilibrium is reached the pressure of CO2 is 0.220 atm. 0.300 atm of CO2 is added, while keeping the temperature constant and the system is allowed to reach again equilibrium. What will be the final mass of CaCO3

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 08:30
Since the gas in your graduated cylinder is a mixture of butane and water vapor, you must determine the partial pressure of the butane, pbutane, alone. to do this, consult a reference and record the partial pressure of the water vapor, pwater, at the temperature you recorded. use the following formula to compute the partial pressure of the butane. pbutane = atmosphere - pwater use the following combined gas law formula and compute the volume that the butane sample will occupy at stp. (hint: convert both temperatures to kelvin.) pbutane x voriginal = pstandard x vfinal troom tstandard use the following ratio and proportion formula to determine the mass of butane needed to occupy a volume of 22.4 l at stp. grams of butane you used “x” grams of butane ml of butane corrected to stp = 22,400 ml compute the theoretical molar mass of butane based on its formula and the atomic masses on the periodic table. compare your experimental results from #3 to the theoretical value of #4, computing a percent error of your findings using this formula: % error = measured value - accepted value x 100 accepted value use the following ratio and proportion formula to determine the mass of butane needed to occupy a volume of 22.4 l at stp. need asap
Answers: 1

Chemistry, 22.06.2019 18:00
An object displaces 652 ml of water. the volume of the object is: 0.652 cm³ 6.52 cm³ 65.2 cm³ 652 cm³
Answers: 3

Chemistry, 23.06.2019 04:10
An unknown substance has been shown to have metallic bonds. which of the following is most likely a property of this substance? a. low conductivity b. low boiling point c. high malleability d. high solubility in water
Answers: 2
You know the right answer?
0.100 mol of CaCO3 and 0.100 mol CaO are placed in an 10.0 L evacuated container and heated to 385 K...
Questions

English, 02.09.2019 00:10

Business, 02.09.2019 00:10




Biology, 02.09.2019 00:10



Biology, 02.09.2019 00:10


English, 02.09.2019 00:10


Mathematics, 02.09.2019 00:10


History, 02.09.2019 00:10

Social Studies, 02.09.2019 00:10

Physics, 02.09.2019 00:10

Physics, 02.09.2019 00:10


Mathematics, 02.09.2019 00:10

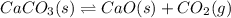
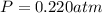
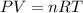 ( ideal gas equation)
( ideal gas equation)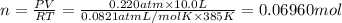
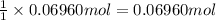 calcium carbonate
calcium carbonate