
What mass of silver chloride can be produced from 1.30 L of a 0.245 M solution of silver nitrate? The reaction described in Part A required 3.36 L of calcium chloride. What is the concentration of this calcium chloride solution?
Please explain, I don't know where to begin.
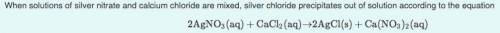

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:00
Explain which group an element with the electron configuration 1s2 2s2 2p6 3s2 3p6 3d1 4s2 belongs to.
Answers: 3

Chemistry, 22.06.2019 09:00
This chart lists four kinds of polymers and their sources. what can be known about all four polymers, despite their differences? they come from living things. they share ionic carbon bonds. they are at least 100 monomers long. they are made of repeating subunits.
Answers: 3

Chemistry, 22.06.2019 09:20
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Chemistry, 22.06.2019 22:00
4.25g sample of solid ammonium nitrate dissolves in 60.0g of water in a coffee-cup calorimeter, the temperature drops from 22.0 c to 16.9 c. assume that the specific heat of the solution is the same as that of pure water. calculate delta(h) (in kj/mol nh4no3) for the solution proces.
Answers: 2
You know the right answer?
What mass of silver chloride can be produced from 1.30 L of a 0.245 M solution of silver nitrate? Th...
Questions

History, 16.07.2021 15:40



Arts, 16.07.2021 15:40




Mathematics, 16.07.2021 15:40

Mathematics, 16.07.2021 15:40

Mathematics, 16.07.2021 15:40












