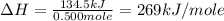Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:20
Concerning the 10.0 ml of 0.50 m nacl to 100 ml of solution: when a solution is diluted, does it change the number of moles dissolved?
Answers: 3


Chemistry, 22.06.2019 11:30
Voltaic cells produce a positive overall charge. what does this indicate? a. the reaction is likely to be endothermic. b. the reaction is spontaneous. c. the reaction is not likely to occur. d. the reaction is not spontaneous.
Answers: 3

Chemistry, 22.06.2019 15:30
Which suspect most likely committed the robbery and how do you know
Answers: 2
You know the right answer?
A bomb calorimeter has a heat capacity of 675 J/°C and contains 925 g of water. If the combustion of...
Questions




Computers and Technology, 02.10.2019 16:00

History, 02.10.2019 16:00


English, 02.10.2019 16:00

Mathematics, 02.10.2019 16:00


Mathematics, 02.10.2019 16:00

Mathematics, 02.10.2019 16:00



Biology, 02.10.2019 16:00

English, 02.10.2019 16:00



History, 02.10.2019 16:00

Biology, 02.10.2019 16:00

History, 02.10.2019 16:00

![q=[q_1+q_2]](/tpl/images/0549/6528/341bc.png)
![q=[c_1\times \Delta T+m_2\times c_2\times \Delta T]](/tpl/images/0549/6528/1d50b.png)
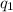 = heat absorbed by the calorimeter
= heat absorbed by the calorimeter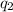 = heat absorbed by the water
= heat absorbed by the water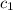 = specific heat of calorimeter =
= specific heat of calorimeter = 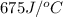
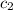 = specific heat of water =
= specific heat of water = 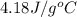
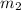 = mass of water = 925 g
= mass of water = 925 g = change in temperature =
= change in temperature = 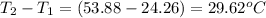
![q=[(675J/^oC\times 29.62^oC)+(925g\times 4.18J/g^oC\times 29.62^oC)]](/tpl/images/0549/6528/f9109.png)
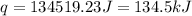
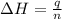
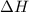 = enthalpy change = ?
= enthalpy change = ?