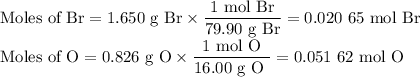Chemistry, 12.03.2020 22:07 jadenyankey
Oxides of virtually every element are known. Bromine, for example, forms several oxides when treated with ozone. Suppose you allow 1.650 g of bromine, Br2, to react with ozone and obtain 2.476 g of BrxOy. What is the formula of the product?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:30
Used the balanced equation 2h2+ o2 - -> 2h2o. if you have 7.2 grams of o2 , how many grams of h2o can you produce ?
Answers: 2

Chemistry, 22.06.2019 15:00
Large helium-filled balloons are used to lift scientific equipment to high altitudes. what is the pressure inside such a balloon if it starts out at sea level with a temperature of 10.0ºc and rises to an altitude where its volume is twenty times the original volume and its temperature is – 50.0ºc ?
Answers: 2


Chemistry, 23.06.2019 00:20
4. propanol and isopropanol are isomers. this means that they have a) the same molecular formula but different chemical properties. b) different molecular formulas but the same chemical properties. c) the same molecular formula and the same chemical properties. d) the same molecular formula but represent different states of the compound
Answers: 3
You know the right answer?
Oxides of virtually every element are known. Bromine, for example, forms several oxides when treated...
Questions


Business, 11.07.2019 00:00



English, 11.07.2019 00:00

Biology, 11.07.2019 00:00


Mathematics, 11.07.2019 00:00



History, 11.07.2019 00:00

Physics, 11.07.2019 00:00


History, 11.07.2019 00:00




Mathematics, 11.07.2019 00:00

Mathematics, 11.07.2019 00:00

Physics, 11.07.2019 00:00