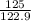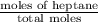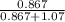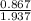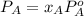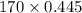Chemistry, 10.03.2020 07:25 starfox5454
At a certain temperature the vapor pressure of pure heptane (C_7 H_16) is measured to be 170. torr. Suppose a solution is prepared by mixing 86.7 g of heptane and 125. g of acetyl bromide (CH_3 COBr). Calculate the partial pressure of heptane vapor above this solution. Round your answer to 3 significant digits. Note for advanced students: you may assume the solution is ideal.

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 15:00
20 pts ‼️ an unmanned spacecraft travels to mars. mars has a lower strength of gravity than earth. where in the image is the spacecraft’s weight the greatest?
Answers: 1

Chemistry, 22.06.2019 15:20
Identify arrows pointing to bonding electrons. done h-0-0-h ) intro
Answers: 3

Chemistry, 22.06.2019 21:30
In science class richard learns that a substance has a boiling point of 230 fahrenheit his teacher ask him to convert this temperature to degrees celsius what is the boiling point of his substance in degrees celsius
Answers: 3
You know the right answer?
At a certain temperature the vapor pressure of pure heptane (C_7 H_16) is measured to be 170. torr....
Questions


Social Studies, 16.10.2020 22:01

Mathematics, 16.10.2020 22:01


Mathematics, 16.10.2020 22:01

Mathematics, 16.10.2020 22:01

Mathematics, 16.10.2020 22:01

English, 16.10.2020 22:01


Mathematics, 16.10.2020 22:01




Mathematics, 16.10.2020 22:01



English, 16.10.2020 22:01

Mathematics, 16.10.2020 22:01


Social Studies, 16.10.2020 22:01

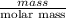
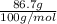
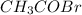 are calculated as follows.
are calculated as follows.