
Chemistry, 10.03.2020 00:00 CarQuestionl6367
Assume that you dissolve 10.1 g of a mixture of NaOH and Ba(OH)2 in 253.0 mL of water and titrate with 1.53 M hydrochloric acid. The titration is complete after 107.8 mL of the acid has been added. What is the mass (in grams) of NaOH in the mixture?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 17:30
How many grams of magnesium metal will react completely with 8.3 liters of 5.5 m hcl? show all of the work needed to solve this problem. mg (s) + 2hcl (aq) → mgcl2 (aq) + h2 (g)
Answers: 3

Chemistry, 23.06.2019 05:30
Awhite powder is added to a solution. the images show observations made before the powder is added, just after the powder has been added, and a little while later. (the liquid in the small beaker is phenol red solution.) what evidence shows that a chemical change has taken place?
Answers: 1

Chemistry, 23.06.2019 06:00
What does it mean for something to be dissolved in watera- it is submerged in water moleculesb-it is stirred in the water moleculesc- it is surrounded by water molecules d-it has water molecules added to it
Answers: 2

Chemistry, 23.06.2019 11:00
The decimals you found in part b are called repeating decimals. in the gizmo, repeating decimals are rounded to two places. how does the gizmo show you that a decimal has been rounded?
Answers: 3
You know the right answer?
Assume that you dissolve 10.1 g of a mixture of NaOH and Ba(OH)2 in 253.0 mL of water and titrate wi...
Questions



History, 21.09.2019 01:30

Mathematics, 21.09.2019 01:30




Biology, 21.09.2019 01:30


Mathematics, 21.09.2019 01:30

Mathematics, 21.09.2019 01:30

History, 21.09.2019 01:30


Social Studies, 21.09.2019 01:30



Biology, 21.09.2019 01:30

Mathematics, 21.09.2019 01:30

Mathematics, 21.09.2019 01:30

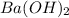 be y.
be y.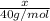
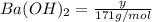
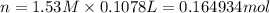
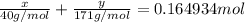
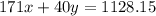 ..[2]
..[2]


