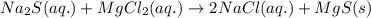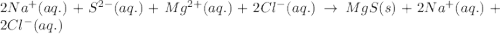Chemistry, 07.03.2020 03:12 foodisbae45678
An aqueous solution of sodium sulfide is allowed to react with an aqueous solution of magnesium chloride. The complete ionic equation contains which of the following species (when balanced in standard form)?a. Cl-(aq)b.2Na+(aq)c.2S2-(aq)d. Na+(aq)e.3Mg2+(aq)

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 08:40
For each of the following compounds, write the formula then predict whether it would be a strong, weak, or non-electrolyte when placed in di water. for the ionic compounds only, put (s) or (aq) after the forrmula formula strong, weak or non electrolyte? a calcium hydroxide b. silver carbonate c. lead(ii) sulfate d. phosphorus trifluoride e. sodium phosphide f barium sulfate g. strontium acetate h. zinc nitrate
Answers: 3

Chemistry, 22.06.2019 19:30
Estimate the molar mass of the gas that effuses at 1.6 times the effusion rate of carbon dioxide.
Answers: 1

Chemistry, 23.06.2019 00:30
If there are 3.5 moles of koh, how many moles of naoh can be produced? question 1 options: a)3.0 moles naoh b)3.5 moles naoh c)1 moles naoh d)9 moles naoh
Answers: 1

Chemistry, 23.06.2019 02:30
Apound is approximately 0.45 kilogram. a persons weighs 87 kilograms. what is the persons’s weight, in pounds, when expressed to the correct number of significant figures
Answers: 1
You know the right answer?
An aqueous solution of sodium sulfide is allowed to react with an aqueous solution of magnesium chlo...
Questions

Mathematics, 18.03.2021 01:10


Mathematics, 18.03.2021 01:10



SAT, 18.03.2021 01:10


Mathematics, 18.03.2021 01:10


Mathematics, 18.03.2021 01:10



Geography, 18.03.2021 01:10




Mathematics, 18.03.2021 01:10

Mathematics, 18.03.2021 01:10


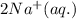 ions
ions