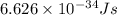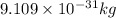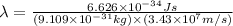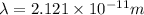Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 20:10
The lattice enthalpy (formation of ionic solid from ions in the gas phase) for agcl(s) is -916 kj/mol and the hydration enthalpy (dissolution of gaseous ions into water) is -850 kj/mol. how much heat (in joules) is involved in forming 1l of saturated agcl solution (1.8 × 10-4 g / 100 ml water) by dissolving agcl(s)? assume solution volume does not change much upon dissolution. the equations are given below. ag+(g) + cl−(g) æ agcl(s)
Answers: 3

Chemistry, 22.06.2019 20:30
We are hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 2


Chemistry, 22.06.2019 22:50
At the current rate, a graph of carbon dioxide produced by fossil fuels over time would slope upward slope downward be horizontal be vertical
Answers: 3
You know the right answer?
What is the wavelength of an electron with a mass of 9.109×10−31 kg and a velocity of 3.43×107 ms? U...
Questions




Mathematics, 02.11.2020 20:10

Mathematics, 02.11.2020 20:10


Mathematics, 02.11.2020 20:10


Mathematics, 02.11.2020 20:10

Advanced Placement (AP), 02.11.2020 20:10

Mathematics, 02.11.2020 20:10

History, 02.11.2020 20:10




Chemistry, 02.11.2020 20:10


Mathematics, 02.11.2020 20:10


Mathematics, 02.11.2020 20:10

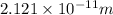
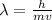
 = De-Broglie's wavelength = ?
= De-Broglie's wavelength = ?