

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 19:00
What information does a complete ionic equation give that the balanced equation doesn’t show?
Answers: 1

Chemistry, 23.06.2019 00:30
Five different substances are given to you to be dissolved in water. which substances are most likely to undergo dissolution in water? check all that apply. view available hint(s) check all that apply. sodium fluoride, naf octane, c8h18 propanol, ch3ch2ch2oh potassium iodide, ki benzene, c6h6
Answers: 1

Chemistry, 23.06.2019 06:10
2. what two items do autotrophs take from the environment to produce their food? 3. what are the two items that are released during transpiration from leaves? 4. what are the two membranes of the system? a.what are the two stages of photosynthesis? what are the two parts of photosynthesis?
Answers: 2

Chemistry, 23.06.2019 08:00
Pl what kind of reaction is this? nahco3 + h2o → co2 + naoh + h2o -composition -decomposition -single replacement -double replacement im leaning more toward single replacement. if im wrong can you explain whyy?
Answers: 1
You know the right answer?
The voltage generated by the zinc concentration cell described by the line notation Zn ( s ) ∣ ∣ Zn...
Questions



Mathematics, 25.03.2021 22:40


Mathematics, 25.03.2021 22:40


Mathematics, 25.03.2021 22:40

Mathematics, 25.03.2021 22:40

Mathematics, 25.03.2021 22:40

Mathematics, 25.03.2021 22:40

Mathematics, 25.03.2021 22:40



Geography, 25.03.2021 22:40


Mathematics, 25.03.2021 22:40

Mathematics, 25.03.2021 22:40

Mathematics, 25.03.2021 22:40


History, 25.03.2021 22:40

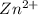 ion at cathode is 0.704 M
ion at cathode is 0.704 M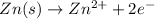
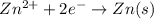
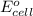 will be equal to zero.
will be equal to zero.![E_{cell}=E^o_{cell}-\frac{0.0592}{n}\log \frac{[Zn^{2+}_{anode}]}{[Zn^{2+}_{cathode}]}](/tpl/images/0536/7525/d92a3.png)
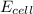 = 25.0 mV = 0.025 V (Conversion factor: 1 V = 1000 mV)
= 25.0 mV = 0.025 V (Conversion factor: 1 V = 1000 mV)![[Zn^{2+}_{cathode}]](/tpl/images/0536/7525/b27eb.png) = ? M
= ? M![[Zn^{2+}_{anode}]](/tpl/images/0536/7525/66f39.png) = 0.100 M
= 0.100 M![0.025=0-\frac{0.0592}{2}\log \frac{0.100}{[Zn^{2+}_{anode}]}](/tpl/images/0536/7525/4f785.png)
![[Zn^{2+}_{anode}]=0.704M](/tpl/images/0536/7525/377d9.png)


