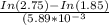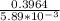Chemistry, 03.03.2020 05:45 Mariela2699
N2O5 decomposes to form NO2 and O2 with first-order kinetics. How long does it take for the N2O5 concentration to decrease from its initial value of 2.75 M to its final value of 1.85 M, if the rate constant, k, equals 5.89 × 10−3?

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 06:40
Which statement correctly describes metallic bonds? a. they form when certain atoms lose electrons and other atoms gain electrons. b. they involve an attraction between anions and cations. they always involvpoth a metal and a nonmetal. d. they can only form between atoms of the same element. e. they form because electrons can move freely between atoms.
Answers: 3


Chemistry, 22.06.2019 20:40
Select the correct value for the indicated bond angle in each of the compounds. o−o−oo−o−o angle of o3 90° 109.5° < 109.5° 120° < 120° 180° f−b−ff−b−f angle of bf3 180° < 109.5° < 120° 120° 109.5° 90° f−o−ff−o−f angle of of2 < 120° 120° 90° 109.5° 180° < 109.5° cl−be−clcl−be−cl angle of becl2 90° 109.5° 180° 120° < 109.5° < 120° f−p−ff−p−f angle of pf3 90° 109.5° < 109.5° 180° 120° < 120° h−c−hh−c−h angle of ch4 90° < 109.5° 180° 120° < 120° 109.5°
Answers: 1
You know the right answer?
N2O5 decomposes to form NO2 and O2 with first-order kinetics. How long does it take for the N2O5 con...
Questions


Mathematics, 15.04.2021 18:20






Mathematics, 15.04.2021 18:20



Mathematics, 15.04.2021 18:20

History, 15.04.2021 18:20

English, 15.04.2021 18:20

Mathematics, 15.04.2021 18:20

History, 15.04.2021 18:20

Mathematics, 15.04.2021 18:20