Chemistry, 02.03.2020 16:51 vladisking888
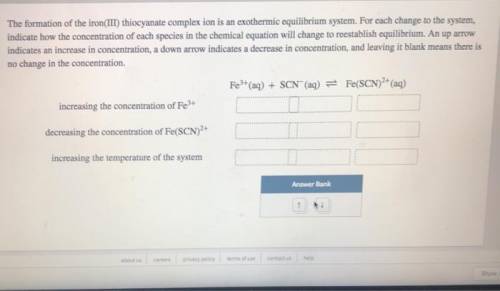
The formation of the iron(III) thiocyanate complex ion is an exothermic equilibrium system. For each change to the system, indicate how the concentration of each species in the chemical equation will change to reestablish equilibrium. An up arrow indicates an increase in concentration, a down arrow indicates a decrease in concentration, and leaving it blank means there is no change in the concentration.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 03:00
Compare the valence electron configuration of the nobles gas elements seen here. what statement is correct?
Answers: 2

Chemistry, 22.06.2019 04:30
How many grams of co(g) are there in 74.5 ml of the gas at 0.933 atm and 30o c?
Answers: 1


Chemistry, 22.06.2019 19:40
What type of electromagnetic waves does the human eye see as the colors red blue or green a visible light waves b radio waves c infrared waves d microwaves
Answers: 1
You know the right answer?
The formation of the iron(III) thiocyanate complex ion is an exothermic equilibrium system. For each...
Questions

Mathematics, 29.10.2020 01:10

Mathematics, 29.10.2020 01:10


History, 29.10.2020 01:10

Biology, 29.10.2020 01:10

History, 29.10.2020 01:10


Mathematics, 29.10.2020 01:10







Mathematics, 29.10.2020 01:10


English, 29.10.2020 01:10