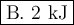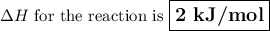Calculate the enthalpy change,,ΔH=∑ Hp - ∑ HR for the following reaction using equations 1, 2 C graphite(s) --> C diamond(s) The following is known. 1. Cgraphite(s) + O2(g) --> CO2(g) ∆H = -394 kJ 2. Cdiamond(s) + O2(g) --> CO2(g) ∆H = -396 kJ A. -763kJ B. 2kJ C. 790kJ D. -2kJ

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:40
22. a flask containing 450 ml of 0.50 m h2so4 was accidentally knocked to the floor. how many grams of nahco, do you need to put on the spill to neutralize the acid according to the following equation: h2so4(aq)+2 nahcos(aq) na,so(aq) +2 h20()+2 co2(g) d) 38 g a) 2.3 g b) 9.5 g c) 19 g
Answers: 1

Chemistry, 22.06.2019 12:30
Acontrol during an experiment. might change remains constant does not exist does change
Answers: 1

Chemistry, 22.06.2019 21:00
Use the measurements in the table to determine which unidentified metal has the highest density. metal volume mass a 10.5 cm3 122 g b 14.2 cm3 132 g c 16.1 cm3 115 g d 12.7 cm3 126 g
Answers: 2

Chemistry, 22.06.2019 22:30
What methods could you use to solubilize calcium carbonate
Answers: 1
You know the right answer?
Calculate the enthalpy change,,ΔH=∑ Hp - ∑ HR for the following reaction using equations 1, 2 C grap...
Questions

History, 08.06.2021 18:50

Mathematics, 08.06.2021 18:50


Biology, 08.06.2021 19:00



Mathematics, 08.06.2021 19:00





Mathematics, 08.06.2021 19:00

Mathematics, 08.06.2021 19:00

Mathematics, 08.06.2021 19:00

Mathematics, 08.06.2021 19:00

Mathematics, 08.06.2021 19:00

History, 08.06.2021 19:00



Mathematics, 08.06.2021 19:00