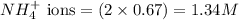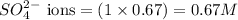Chemistry, 29.02.2020 01:36 kcombest7219
Calculate the concentration of all ions present in each of the following solutions of strong electrolytes. a. 0.0200 mol sodium phosphate in 10.0 mL of solution. 0.300 mol of barium nitrate in 600.0 mL of solution. 1.00 g of potassium chloride in 0.500 L of solution. 132 g of ammonium sulfate in 1.50 L of solution

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 03:00
Which of these would be caused by a chemical change? a) the formation of lava. b) sedimantary rock layering over time. c) metamorphic rock forming from igneous. d) metamorphic rock eroding to form sedimentary rock.
Answers: 3

Chemistry, 22.06.2019 05:50
According to coulomb's law, how would the electrical force between particles change if the product of their electrical charge increased?
Answers: 1

Chemistry, 22.06.2019 12:00
1. if you have a gas at 127 degrees c, what is it's absolute temperature (kelvin)? a. 200kb. 300kc. 400kd. 500k2. if you had a gas whose absolute temperature measured 45 k, what is that temperature in celsius? a. -228 cb. -300 cc. 125 cd. 112 c
Answers: 2

Chemistry, 22.06.2019 15:30
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 1
You know the right answer?
Calculate the concentration of all ions present in each of the following solutions of strong electro...
Questions


Physics, 14.10.2019 06:30

Biology, 14.10.2019 06:30


Social Studies, 14.10.2019 06:30

English, 14.10.2019 06:30

Social Studies, 14.10.2019 06:30


History, 14.10.2019 06:30

History, 14.10.2019 06:30

Spanish, 14.10.2019 06:30


Health, 14.10.2019 06:30



Mathematics, 14.10.2019 06:30




Mathematics, 14.10.2019 06:30

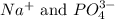 ions in the solution are 6 M and 2 M respectively.
ions in the solution are 6 M and 2 M respectively.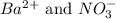 ions in the solution are 0.5 M and 1.0 M respectively.
ions in the solution are 0.5 M and 1.0 M respectively.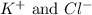 ions in the solution are 0.051 M and 0.051 M respectively.
ions in the solution are 0.051 M and 0.051 M respectively.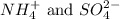 ions in the solution are 1.34 M and 0.67 M respectively.
ions in the solution are 1.34 M and 0.67 M respectively.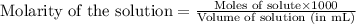 ......(1)
......(1)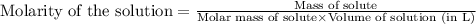 ...(2)
...(2)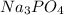
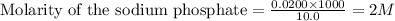
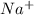 ions and 1 mole of
ions and 1 mole of 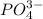 ions
ions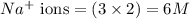
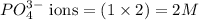
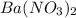
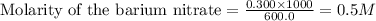
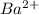 ions and 2 mole of
ions and 2 mole of 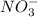 ions
ions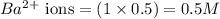
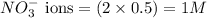
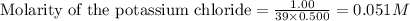
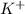 ions and 1 mole of
ions and 1 mole of 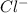 ions
ions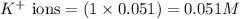
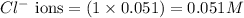
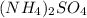
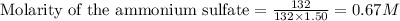
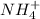 ions and 1 mole of
ions and 1 mole of 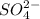 ions
ions