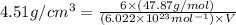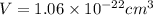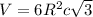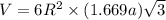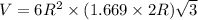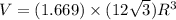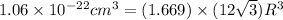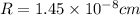Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 17:10
Nitric oxide (no) can be formed from nitrogen, hydrogen and oxygen in two steps. in the first step, nitrogen and hydrogen react to form ammonia: n2(g) + 2 h_2(g) rightarrow 2 nh_3 (g) delta h = -92. kj in the second step, ammonia and oxygen react to form nitric oxide and water: 4 nh_3(g) + 5 o_2(g) rightarrow 4no(g) + 6 h_2o(g) delta h = -905. kj calculate the net change in enthalpy for the formation of one mole of nitric oxide from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 1

Chemistry, 21.06.2019 17:50
Which best describes why nh4+ can form an ionic bond with cl-?
Answers: 3

Chemistry, 22.06.2019 00:00
Which of the following methods uses the decay of atomic particles in an object to find its exact age? a. fossil dating b. geologic dating c. radioactive dating d. relative dating
Answers: 1

You know the right answer?
Titanium has an HCP crystal structure, a c/a ratio of 1.669, an atomic weight of 47.87 g/mol, and a...
Questions

Mathematics, 02.08.2019 05:50



Mathematics, 02.08.2019 05:50


Mathematics, 02.08.2019 05:50


Mathematics, 02.08.2019 05:50


Mathematics, 02.08.2019 05:50


English, 02.08.2019 05:50






Mathematics, 02.08.2019 05:50


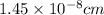
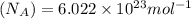
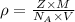 .............(1)
.............(1) = density =
= density = 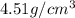
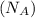 = Avogadro's number
= Avogadro's number