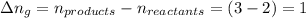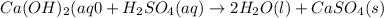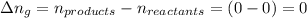Chemistry, 25.02.2020 03:04 DaFuzzyDude
The enthalpy change, ΔH, for a reaction at constant pressure is defined as: ΔH = ΔE + PΔV. For which of the following reactions will ΔH be approximately equal to ΔE? Select all that apply. Group of answer choices 2 NO2(g) -> N2(g) + 2 O2(g)

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:30
Identify the missing numbers below to show the result of multiplying the numbers (1.6 × 10-19)(5.0 × 106) = c × 10d
Answers: 1


Chemistry, 22.06.2019 19:00
Structure of the atoms: discovery of the nucleus in 1909i need answering all of these questions
Answers: 3

Chemistry, 22.06.2019 23:00
Arectangle has a diagonal 20 inches long that forms angles of 60 and 30 with the sides. find the perimeter of the rectangle. for geometry
Answers: 3
You know the right answer?
The enthalpy change, ΔH, for a reaction at constant pressure is defined as: ΔH = ΔE + PΔV. For which...
Questions


Computers and Technology, 09.01.2020 00:31




English, 09.01.2020 00:31





Mathematics, 09.01.2020 00:31









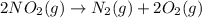
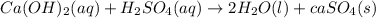
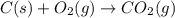
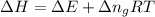 as
as 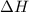 = enthalpy change
= enthalpy change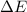 = internal energy change
= internal energy change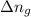 = change in number of moles of gas particles =
= change in number of moles of gas particles =