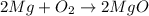Chemistry, 25.02.2020 03:04 tonimgreen17p6vqjq
A student burned 0.250 g of magnesium metal in an open crucible. Oxygen from the air combined with the metal forming magnesium oxide. If 0.421 g of magnesium oxide were produced, what mass of oxygen combined with the metal? 1. 0.171 g 2. 0.671 g 3. 1.684 g 4. 0.594 g

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:30
How many moles are in 250 grams of tungsten (w)? * 4.4x10^23 moles 4.2x10^23 moles 0.7 moles 1.4 moles
Answers: 3

Chemistry, 22.06.2019 13:20
Can someone me with 3 and 4 plz. this is for masteries test.
Answers: 2

Chemistry, 22.06.2019 18:00
What amount of heat is exchanged when 106.2 grams of substance y goes from a liquid at 35 degrees celsius to a solid at the same temperature? melting point of substance y = 35 degrees c; δhvaporization = 3.67 j/mol; δhfusion = 3.30 j/mol. mwsubstance y = 28.22 g/mol. −12.4 j −3.51 x 102 j 1.24 x 101 j 351 j
Answers: 1

Chemistry, 22.06.2019 18:00
Which three statements represent the benefits of performing experiments using computer simulations?
Answers: 2
You know the right answer?
A student burned 0.250 g of magnesium metal in an open crucible. Oxygen from the air combined with t...
Questions



Social Studies, 05.12.2021 20:30


Mathematics, 05.12.2021 20:30

Mathematics, 05.12.2021 20:30

Mathematics, 05.12.2021 20:30

Computers and Technology, 05.12.2021 20:30



English, 05.12.2021 20:40

Chemistry, 05.12.2021 20:40






Biology, 05.12.2021 20:40