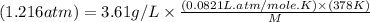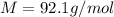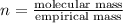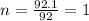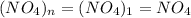Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:00
In a sample of oxygen gas at room temperature, the average kinetic energy of all the balls stays constant. which postulate of kinetic molecular theory best explains how this is possible? a. attractive forces between gas particles are negligible because the particles of an ideal gas are moving so quickly. b. collisions between gas particles are elastic; there is no net gain or loss of kinetic energy. c. gases consist of a large number of small particles, with a lot of space between the particles. d. gas particles are in constant, random motion, and higher kinetic energy means faster movement.
Answers: 1


Chemistry, 22.06.2019 21:30
Harry lives in a city, and he has a lung condition known as asthma. on certain days, harry has to stay inside because pollutants in the air make it difficult for him to breathe. which of these pollution sources are nonpoint sources that might bother harry if he goes outside? choose the two that apply.
Answers: 3

Chemistry, 22.06.2019 22:30
Molecular iodine, i2(g), dissociates into iodine atoms at 625 k with a first-order rate constant of 0.271 s−1. part a part complete what is the half-life for this reaction?
Answers: 3
You know the right answer?
A compound contains only nitrogen and oxygen and is 30.5% nitrogen by mass. A gaseous sample of the...
Questions

Mathematics, 14.04.2020 05:33

Chemistry, 14.04.2020 05:33

Mathematics, 14.04.2020 05:33


Mathematics, 14.04.2020 05:33

English, 14.04.2020 05:33


Mathematics, 14.04.2020 05:34





Mathematics, 14.04.2020 05:34

Mathematics, 14.04.2020 05:34






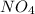
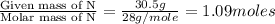
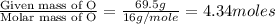
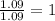
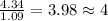
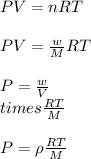
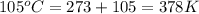
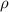 = density of gas = 3.61 g/L
= density of gas = 3.61 g/L