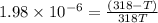Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 12:00
What does a complete balanced chemical equation include? a. exothermic coefficients b. endothermic coefficients c. valence electrons d. molar coefficients
Answers: 1


Chemistry, 22.06.2019 22:30
Which of the following is true about the speed of light? it depends on the wavelength.
Answers: 3

Chemistry, 23.06.2019 00:00
How many moles of water are created if 3 moles of hydrogen react completely with excess oxygen?
Answers: 3
You know the right answer?
A certain liquid has a vapor pressure of 92.0 Torr at 23.0 ∘ C and 378.0 Torr at 45.0∘C.
A. C...
A. C...
Questions



English, 13.11.2020 17:40


Mathematics, 13.11.2020 17:40



Mathematics, 13.11.2020 17:40


Mathematics, 13.11.2020 17:40

Health, 13.11.2020 17:40


Social Studies, 13.11.2020 17:40

English, 13.11.2020 17:40

Chemistry, 13.11.2020 17:40



Mathematics, 13.11.2020 17:40


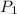 = 92.0 torr,
= 92.0 torr, 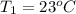 = (23 + 273)K = 296 K
= (23 + 273)K = 296 K
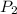 = 378.0 torr,
= 378.0 torr, 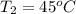 = (45 + 273)K = 318 K
= (45 + 273)K = 318 K
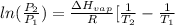
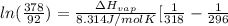
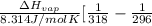
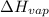 = 2926063.008 J/mol
= 2926063.008 J/mol
![ln (\frac{760 torr}{378 torr}) = -\frac{2926063.008 J/mol }{8.314 J/mol K} [\frac{1}{T} - \frac{1}{318}]](/tpl/images/0508/2864/327ff.png)