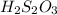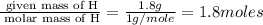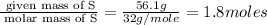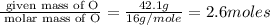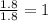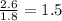A laboratory analysis of a 100 g sample finds it is composed of 1.8 g hydrogen, 56.1 g sulfur, and 42.1 g oxygen. What is its empirical formula? Give your answer in the form H#S#O#, where the number following the element’s symbol corresponds to the subscript in the formula. (Don’t include a 1 subscript explicitly.) For example, the formula CHO would be entered as CH2O.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 03:30
If you have 5.25 grams of methane (ch4), how many grams of co2 will you produce ?
Answers: 1

Chemistry, 23.06.2019 03:30
If you need to add 27.50ml of a solution, which piece of glassware would you use to deliver this volume and explain how you would determine if the 27.50 ml was measured?
Answers: 2

Chemistry, 23.06.2019 08:00
How many distinct monochlorinated products, including stereoisomers, can result when the alkane below is heated in the presence of cl2? 3 4 5 6 7?
Answers: 3

Chemistry, 23.06.2019 15:00
What do we call the rows on the periodic table? a. periodb. familyc. groupd. metals
Answers: 1
You know the right answer?
A laboratory analysis of a 100 g sample finds it is composed of 1.8 g hydrogen, 56.1 g sulfur, and 4...
Questions


Mathematics, 03.03.2021 07:00

Mathematics, 03.03.2021 07:00



English, 03.03.2021 07:00

History, 03.03.2021 07:00

Mathematics, 03.03.2021 07:00



Mathematics, 03.03.2021 07:00





Chemistry, 03.03.2021 07:00


Social Studies, 03.03.2021 07:00