Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 06:00
If a polyatomic ionic compound has gained two hydrogen ions, then how does its name begin?
Answers: 3

Chemistry, 22.06.2019 16:30
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2


Chemistry, 22.06.2019 22:30
Draw the aromatic compound toluene (methylbenzene). show all hydrogen atoms, including those on the ring.
Answers: 1
You know the right answer?
A solution made by dissolving 33 mg of insulin in 6.5 mL of water has an osmotic pressure of 15.5 mm...
Questions



History, 21.07.2019 01:20

Arts, 21.07.2019 01:20

Biology, 21.07.2019 01:20





Biology, 21.07.2019 01:20

Mathematics, 21.07.2019 01:20




History, 21.07.2019 01:20


Mathematics, 21.07.2019 01:20



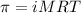
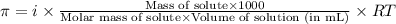
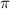 = osmotic pressure of the solution = 15.5 mmHg
= osmotic pressure of the solution = 15.5 mmHg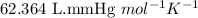
![25^oC=[273+25]=298K](/tpl/images/0508/1937/6a9f9.png)