
Chemistry, 19.12.2019 20:31 harshakayla02
Ascientist measures the standard enthalpy change for the following reaction to be -190.0 kj : hcl(g) + nh3(g) nh4cl(s) based on this value and the standard enthalpies of formation for the other substances, the standard enthalpy of formation of nh4cl(s) is kj/mol.

Answers: 3


Another question on Chemistry



Chemistry, 23.06.2019 01:30
Polar bears give birth and hunt on sea ice. which of the following would polar bears survive during the melting of arctic ice? growing another layer of fur during summer migrate inland to search for different food sources staying put until the ice refreezes sticking to the usual diet of seals
Answers: 1

Chemistry, 23.06.2019 03:00
In november 1987, a massive iceberg broke loose from the antartic ice mass and floated free in the ocean. the chunk of ice was estimated to be 98 mi long, 25 mi wide, and 750 ft thick. a typical backyard swimming pool contains about 24,000 gallons of water. how many of these pools could you fill from the water in this iceberg? (assume the iceberg is a rectangular solid of the above dimensions and consists of water only). express answer in scientific notation.
Answers: 1
You know the right answer?
Ascientist measures the standard enthalpy change for the following reaction to be -190.0 kj : hcl(g...
Questions

English, 15.12.2020 01:00

Biology, 15.12.2020 01:00

History, 15.12.2020 01:00

History, 15.12.2020 01:00

Mathematics, 15.12.2020 01:00

Mathematics, 15.12.2020 01:00


Mathematics, 15.12.2020 01:00

Mathematics, 15.12.2020 01:00

Mathematics, 15.12.2020 01:00

Biology, 15.12.2020 01:00



Computers and Technology, 15.12.2020 01:00



Mathematics, 15.12.2020 01:00



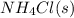 is -328.4 kJ/mol
is -328.4 kJ/mol![\Delta H_{rxn}=\sum [n\times \Delta H_f_{(products)}]-\sum [n\times \Delta H_f_{(reactants)}]](/tpl/images/0426/4202/192b9.png)
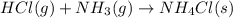
![\Delta H_{rxn}=[(1\times \Delta H_f_{(NH_4Cl(s))})]-[(1\times \Delta H_f_{(NH_3(g))})+(1\times \Delta H_f_{(HCl(g))})]](/tpl/images/0426/4202/589ac.png)
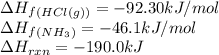
![-190.0=[(1\times \Delta H_f_{(NH_4Cl(s))})]-[(1\times (-46.1))+(1\times (-92.30))]\\\\\Delta H_f_{(NH_4Cl(s))}=-328.4kJ/mol](/tpl/images/0426/4202/9648b.png)



