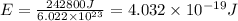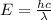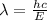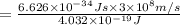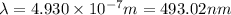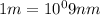Chemistry, 12.12.2019 05:31 carlosiscr7
Acommon way of initiating certain chemical reactions with light involves the generation of free halogen atoms in solution. if δh for the reaction cl 2(g) → 2cl(g) is 242.8 kj/mol, what is the longest wavelength of light that will produce free chlorine atoms in solution?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 13:00
Pauling and lewis questioned the extreme definitions of bonds. they wondered if bonds might be described somewhere in between the two extremes (covalent and ionic). on the basis of experimental data,pauling confirmed that bonds could be ionic, covalent, and for those, in between, exhibit a degree of ionic character. he theorized that the major factor was how strongly the atoms in the bond attracted the electrons. pauling called this factor - the tendency of an atom to attract electrons in a bond.
Answers: 2

Chemistry, 21.06.2019 22:30
1.aluminum chloride (alcl3), and sodium hydroxide (naoh) can react to form aluminum hydroxide (al(oh)3) and sodium chloride (nacl). you have 13.4 g of aluminum chloride and 10.0 g of sodium hydroxide. answer the following questions: •what is the balanced equation for this reaction? •if you use all 13.4 g of aluminum chloride, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •if you use all 10.0 g of sodium hydroxide, how many grams of aluminum hydroxide can be formed? work must be shown to earn credit •how many grams of aluminum hydroxide will actually be made? which reagent is limiting? explain your answer.
Answers: 1

Chemistry, 22.06.2019 12:10
Building glycogen from glucose molecules is an example of
Answers: 3

Chemistry, 22.06.2019 21:30
What is another way to determine mass times acceleration?
Answers: 1
You know the right answer?
Acommon way of initiating certain chemical reactions with light involves the generation of free halo...
Questions

History, 03.07.2019 17:30



Social Studies, 03.07.2019 17:30

Mathematics, 03.07.2019 17:30

History, 03.07.2019 17:30

History, 03.07.2019 17:30

Chemistry, 03.07.2019 17:30

Mathematics, 03.07.2019 17:30

Mathematics, 03.07.2019 17:30

Mathematics, 03.07.2019 17:30

Mathematics, 03.07.2019 17:30

Mathematics, 03.07.2019 17:30

Mathematics, 03.07.2019 17:30




Chemistry, 03.07.2019 17:30


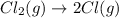 ,ΔH = 242.8 kJ/mol
,ΔH = 242.8 kJ/mol