
Chemistry, 05.12.2019 05:31 sumayyahjj
How much heat (in kj) is required to warm 13.0 g of ice, initially at -10.0 ∘c, to steam at 111.0 ∘c? the heat capacity of ice is 2.09 j/g⋅∘c and that of steam is 2.01 j/g⋅∘c, the heat of fusion for water is 6.02 kj/mol, and the heat of vaporization for water is 40.7 kj/mol.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 03:00
What happened in 2012 and how does it illustrate the importance of understanding the sun and how it works?
Answers: 3

Chemistry, 22.06.2019 03:00
Zoe is investigating the composition of substance a, an unknown substance. using chemical processes, she analyzes substance a and determines it is composed of sodium, oxygen, and hydrogen atoms in a ratio of 1 : 1 : 1. what is substance a? a. a compound b. an element c. a heterogeneous mixture d. a homogeneous mixture
Answers: 1

Chemistry, 22.06.2019 09:40
Consider this initial-rate data at a certain temperature for the reaction described by
Answers: 1

Chemistry, 22.06.2019 21:30
Liquid ammonia is produced at high temperatures and under great pressure in a tank by passing a mixture of nitrogen gas and hydrogen gas over an iron catalyst. the reaction is represented by the following equation. n2(g) + 3h2(g) → 2nh3(g) changing all but one experimental condition will affect the amount of ammonia produced. that condition is a) increasing the concentration of both reactants b) changing the temperature within the tank c) decreasing the pressure within the tank. d) increasing only the amount of nitrogen present.
Answers: 1
You know the right answer?
How much heat (in kj) is required to warm 13.0 g of ice, initially at -10.0 ∘c, to steam at 111.0 ∘c...
Questions


History, 20.07.2019 03:30


Mathematics, 20.07.2019 03:30






Geography, 20.07.2019 03:30


Mathematics, 20.07.2019 03:30


Social Studies, 20.07.2019 03:30


Chemistry, 20.07.2019 03:30

Chemistry, 20.07.2019 03:30



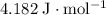 , the melting point of water is
, the melting point of water is 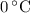 , and that the boiling point of water is
, and that the boiling point of water is 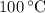 .
.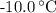 ice to steam at
ice to steam at 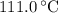 .
.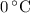 . The change in temperature would be
. The change in temperature would be 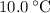 .Step two: supply the heat of fusion to convert that 13.0 gram of ice to water.Step three: heat the 13.0 gram of water from
.Step two: supply the heat of fusion to convert that 13.0 gram of ice to water.Step three: heat the 13.0 gram of water from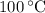 . The change in temperature would be
. The change in temperature would be 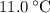 .Energy required for step one, three, and five
.Energy required for step one, three, and five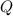 required to raise the temperature of an object by a
required to raise the temperature of an object by a  :
: 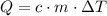 .
.  is the specific heat of this substance,
is the specific heat of this substance, 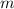 is the mass of the substance, and
is the mass of the substance, and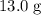 .
.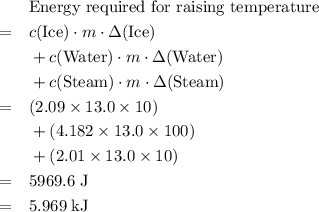 .
.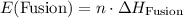 .
.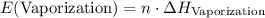 .
.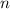 is the number of moles of the substance.
is the number of moles of the substance.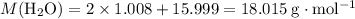 .
.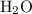 molecules in
molecules in 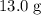 :
: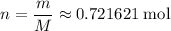 .
. 
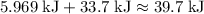 .
.

