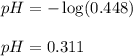Chemistry, 20.11.2019 23:31 infoneetusinghoyg22o
27.3 ml sample of a 0.488 m aqueous hydrofluoric acid solution is titrated with a 0.367 m aqueous potassium hydroxide solution. what is the ph at the start of the titration, before any potassium hydroxide has been added?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:00
Matches the chemical name of each oxide of phosphorus to its chemical formula
Answers: 2

Chemistry, 22.06.2019 08:40
For each of the following compounds, write the formula then predict whether it would be a strong, weak, or non-electrolyte when placed in di water. for the ionic compounds only, put (s) or (aq) after the forrmula formula strong, weak or non electrolyte? a calcium hydroxide b. silver carbonate c. lead(ii) sulfate d. phosphorus trifluoride e. sodium phosphide f barium sulfate g. strontium acetate h. zinc nitrate
Answers: 3

Chemistry, 22.06.2019 13:30
Which statements are true concerning mineral formation? check all that apply. the slower the cooling, the larger the crystals. the faster the cooling, the smaller the crystals. crystals formed from magma are smaller than crystals formed from lava. minerals can only form in solutions when the solution is heated deep underground. when a solution cools, elements and compounds leave the solution and crystallize as minerals. minerals formed from hot water solutions can form narrow channels in the surrounding rock.
Answers: 1

Chemistry, 22.06.2019 13:30
How many protons, electrons, and neutrons are in each of the following isotopes? a. zirconium-90 b. palladium-108 c. bromine-81 d. antimony-123
Answers: 1
You know the right answer?
27.3 ml sample of a 0.488 m aqueous hydrofluoric acid solution is titrated with a 0.367 m aqueous po...
Questions



English, 24.06.2019 18:00

Biology, 24.06.2019 18:00




Advanced Placement (AP), 24.06.2019 18:00












![pH=-\log[H^+]](/tpl/images/0383/5176/cf945.png)
![[H^+]=0.488M](/tpl/images/0383/5176/73a1e.png)