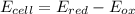Chemistry, 11.11.2019 23:31 mariamakonteh31
In the activity, click on the e∘cell and keq quantities to observe how they are related. use this relation to calculate keq for the following redox reaction that occurs in an electrochemical cell having two electrodes: a cathode and an anode. the two half-reactions that occur in the cell are
cu2+(aq)+2e−→cu(s) and co(s)→co2+(aq)+2e−
the net reaction is
cu2+(aq)+co(s)→cu(s)+co2+(aq)
use the given standard reduction potentials in your calculation as appropriate. ( keq=5.88*10^20)
in the activity, click on the keq and δg∘ quantities to observe how they are related.
calculate δg∘ using this relationship and the equilibrium constant (keq) obtained in part a at t=298k: keq=5.88*10^20
express the gibbs free energy (δg∘) in joules to three significant figures.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:10
Which of these conditions most likely produces an unstable isotope?
Answers: 2

Chemistry, 22.06.2019 08:00
Which of the following observations indicates that there is a small, dense, positively charged part in the center of an atom? some uncharged particles are scattered by a gold foil. all uncharged particles are attracted towards a gold foil. all positively charged particles pass straight through a gold foil. some positively charged particles bounce back from a gold foil.
Answers: 2

Chemistry, 22.06.2019 14:10
Precision can be defined as the o exact center of a data set. o reproducibility of a measured value. o correlation between two variables that are measured in a data set agreement between a measured value and an accepted value.
Answers: 2

Chemistry, 22.06.2019 14:50
Which of the following is most likely true about water in chemical systems? a) water dissolves nonpolar ionic compounds. b) water dissociates ionic compounds. c) water dissociates covalent molecules. d) water dissolves nonpolar covalent substances.
Answers: 1
You know the right answer?
In the activity, click on the e∘cell and keq quantities to observe how they are related. use this re...
Questions


Mathematics, 24.11.2020 19:30


Mathematics, 24.11.2020 19:30

Physics, 24.11.2020 19:30




Biology, 24.11.2020 19:30

Physics, 24.11.2020 19:30



Mathematics, 24.11.2020 19:30



Mathematics, 24.11.2020 19:30

Mathematics, 24.11.2020 19:30


History, 24.11.2020 19:30