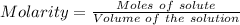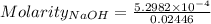Chemistry, 16.10.2019 01:30 jakails828
The concentration of a certain sodium hydroxide solution was determined by using the solution to titrate a sample of potassium hydrogen phthalate (abbreviated as khp). khp is an acid with one acidic hydrogen and a molar mass of 204.22 g/mol. in the titration, 24.46 ml of the sodium hydroxide solution was required to react with 0.1082 g khp. calculate the molarity of the sodium hydroxide.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 07:30
Plz mark brainliest 30 points 1) find the momentum of a 12 kg snowball that is rolling with a velocity of 9 m/s. 2) an 8 ball with a mass of .5 kg is sitting at rest. it is hit by the cue ball (1 kg) traveling at 2.5 m/s. if the cue ball is at rest after the collision, how fast is the 8 ball traveling after the collision? 3) two football players are running toward each other. if the offensive player is 75 kg and is running 8 m/s, how fast must the 60 kg defensive player run in order for the two players to hit and stop?
Answers: 1

Chemistry, 22.06.2019 13:30
Why does asexual reproduction result in offspring with identicle genetic variation
Answers: 2

Chemistry, 22.06.2019 22:30
What methods could you use to solubilize calcium carbonate
Answers: 1

Chemistry, 23.06.2019 01:00
If a straight-chain hydrocarbon is a gas at room temperature, how many carbon atoms will it have? a. 6 carbon atoms b. 12 carbon atoms c. 24 carbon atoms d. 3 carbon atoms
Answers: 1
You know the right answer?
The concentration of a certain sodium hydroxide solution was determined by using the solution to tit...
Questions

Biology, 10.05.2021 21:30



Mathematics, 10.05.2021 21:30

World Languages, 10.05.2021 21:30

History, 10.05.2021 21:30


English, 10.05.2021 21:30




English, 10.05.2021 21:30

Mathematics, 10.05.2021 21:30


English, 10.05.2021 21:30




Mathematics, 10.05.2021 21:30

English, 10.05.2021 21:30

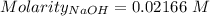
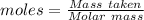
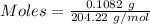
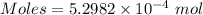
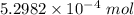 of KHP reacts with
of KHP reacts with