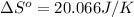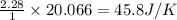Chemistry, 02.10.2019 21:30 dontworry48
Consider the reaction: h2(g) + cl2(g) --> 2hcl(g)
using standard absolute entropies at 298k, calculate the entropy change for the system when 2.28 moles of h2(g)react at standard conditions.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 01:30
Arollercoaster car at the top of a hill has potential energy kinetic energy chemical energy light energy
Answers: 1


Chemistry, 22.06.2019 09:50
Although respiratory organs vary across different organisms, they all contain respiratory surfaces that have a large surface area and are extremely thin. explain why having an extremely thin respiratory surface with a large surface area is advantageous for the process of gas exchange
Answers: 1

You know the right answer?
Consider the reaction: h2(g) + cl2(g) --> 2hcl(g)
using standard absolute entropies...
using standard absolute entropies...
Questions


Social Studies, 04.12.2019 21:31


Mathematics, 04.12.2019 21:31

English, 04.12.2019 21:31






Mathematics, 04.12.2019 21:31

Social Studies, 04.12.2019 21:31

Chemistry, 04.12.2019 21:31

Social Studies, 04.12.2019 21:31

History, 04.12.2019 21:31

History, 04.12.2019 21:31


Arts, 04.12.2019 21:31


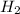 reacts at standard condition is 45.8 J/K
reacts at standard condition is 45.8 J/K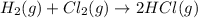
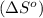 is:
is: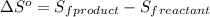
![\Delta S^o=[n_{HCl}\times \Delta S_f^0_{(HCl)}]-[n_{H_2}\times \Delta S_f^0_{(H_2)}+n_{Cl_2}\times \Delta S_f^0_{(Cl_2)}]](/tpl/images/0284/2286/d7d45.png)
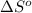 = entropy change of reaction = ?
= entropy change of reaction = ?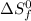 = standard entropy of formation
= standard entropy of formation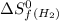 = 130.684 J/mol.K
= 130.684 J/mol.K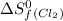 = 223.066 J/mol.K
= 223.066 J/mol.K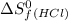 = 186.908 J/mol.K
= 186.908 J/mol.K![\Delta S^o=[2mole\times (186.908J/K.mole)]-[1mole\times (130.684J/K.mole)+1mole\times (223.066J/K.mole)}]](/tpl/images/0284/2286/55b6e.png)