Option c
Explanation:
In the allene, 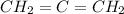
Central atom forms two sigma bond and two pi bond.
Hybridization of central atom = sp
Hybridization of two terminal C = 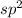
Allene is a nonplanar molecules. This is because of the planes of two 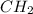 groups which are twisted 90 ° to each other.
groups which are twisted 90 ° to each other.
Therefore, the two H atoms on one C lie above and below the plane defined by the other 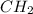 group.
group.


























