
Chemistry, 02.09.2019 22:20 Jackiecroce12
H2 is produced by the reaction of 118.5 ml of a 0.8775-m solution of h3po4 according to the following equations: 2cr +
2h3po4 ? 3h2 + 2crpo4.
(a) outline the steps neccessary to determine the number of moles and mass of h2
(b)perform the calculations outlined

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 03:00
How does a hydroelectric power plant converts energy into energy.
Answers: 1

Chemistry, 22.06.2019 09:00
Particles vibrate in a rigid structure and do not move relative to their neighbors.
Answers: 1

Chemistry, 22.06.2019 10:00
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1

Chemistry, 22.06.2019 12:20
Consider the reaction of a(g) + b(g) + c(g) => d(g) for which the following data were obtained: experiment initial [a], mol/l initial [b], mol/l initial [c], mol/l initial rate, mol/l.s 1 0.0500 0.0500 0.0100 6.25 x 10^-3 2 0.100 0.0500 0.0100 2.50 x 10^-2 3 0.100 0.100 0.0100 1.00 x 10^-1 4 0.0500 0.0500 0.0200 6.25 x 10^-3 what is the rate law for the reaction?
Answers: 3
You know the right answer?
H2 is produced by the reaction of 118.5 ml of a 0.8775-m solution of h3po4 according to the followin...
Questions



Mathematics, 30.06.2019 17:40

Social Studies, 30.06.2019 17:40


History, 30.06.2019 17:40




Mathematics, 30.06.2019 17:50

Social Studies, 30.06.2019 17:50

Mathematics, 30.06.2019 17:50

Social Studies, 30.06.2019 17:50

Health, 30.06.2019 17:50

History, 30.06.2019 17:50




Computers and Technology, 30.06.2019 17:50

History, 30.06.2019 17:50

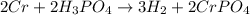
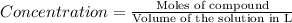
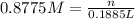
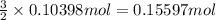 of hydrogen gas
of hydrogen gas

