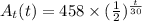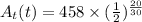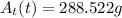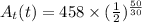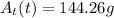Chemistry, 19.08.2019 18:20 saabrrinnaaa
The radioactive substance cesium-137 has a half-life of 30 years. the amount at (in grams) of a sample of cesium-137 remaining after t years is given by the following exponential function.
at(t)=458(1/2)^(t/30)
find the amount of the sample remaining after 20 years and after 50 years. round your answers to the nearest gram as necessary.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:20
Calcium hydride (cah2) reacts with water to form hydrogen gas: cah2(s) + 2h2o(l) → ca(oh)2(aq) + 2h2(g) how many grams of cah2 are needed to generate 45.0 l of h2 gas at a pressure of 0.995 atm and a temperature of 32 °c?
Answers: 2

Chemistry, 22.06.2019 02:40
How many liters of hydrogen gas will be produced at stp from the reaction of 7.179×10^23 atoms of magnesium with 54.219g of phosphoric acid (h3po4) the equation is 3mg + 2h3(> mg(po4)2+3h2
Answers: 1

Chemistry, 22.06.2019 05:30
What type of reaction is shown below? check all that apply. 2h2o2 → 2h2o + o2 synthesis decomposition combustion
Answers: 1

Chemistry, 22.06.2019 14:30
Aroom with dimensions 7.00m×8.00m×2.50m is to be filled with pure oxygen at 22.0∘c and 1.00 atm. the molar mass of oxygen is 32.0 g/mol. how many moles noxygen of oxygen are required to fill the room? what is the mass moxygen of this oxygen?
Answers: 1
You know the right answer?
The radioactive substance cesium-137 has a half-life of 30 years. the amount at (in grams) of a samp...
Questions

English, 05.01.2020 08:31


Social Studies, 05.01.2020 08:31


Mathematics, 05.01.2020 08:31

Mathematics, 05.01.2020 08:31

English, 05.01.2020 08:31

Mathematics, 05.01.2020 08:31

English, 05.01.2020 08:31


Biology, 05.01.2020 08:31


History, 05.01.2020 08:31



Mathematics, 05.01.2020 08:31

History, 05.01.2020 08:31


Mathematics, 05.01.2020 08:31

Social Studies, 05.01.2020 08:31