

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:00
The pressure inside a hydrogen-filled container was 2.10 atm at 21 ? c. what would the pressure be if the container was heated to 92 ? c ?
Answers: 2

Chemistry, 22.06.2019 08:40
Which statement can best be concluded from the ideal gas law?
Answers: 2


Chemistry, 22.06.2019 22:30
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3
You know the right answer?
The (oh) of a given solution =1.00x10-9m. what is this solution s poh?...
Questions

Mathematics, 20.09.2020 05:01


History, 20.09.2020 05:01



Spanish, 20.09.2020 05:01

Mathematics, 20.09.2020 05:01


Mathematics, 20.09.2020 05:01

Advanced Placement (AP), 20.09.2020 05:01


Mathematics, 20.09.2020 05:01

Mathematics, 20.09.2020 05:01

Mathematics, 20.09.2020 05:01

Mathematics, 20.09.2020 05:01

Health, 20.09.2020 05:01



Mathematics, 20.09.2020 05:01

Mathematics, 20.09.2020 05:01

![pOH=-log[OH^-]](/tpl/images/0171/9043/4fd6b.png)
![pOH=-log[1.00\times10^{-9}M]](/tpl/images/0171/9043/de785.png)
![=-[-9]](/tpl/images/0171/9043/92cc9.png)
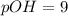
![[OH^-]](/tpl/images/0171/9043/b2910.png) will take place and this will decrease the Hydrogen ion concentration
will take place and this will decrease the Hydrogen ion concentration ![[H^+ ]=[OH^- ]](/tpl/images/0171/9043/9cdae.png)
![[H^+ ][OH^- ]](/tpl/images/0171/9043/24064.png)
![[H^+ ]](/tpl/images/0171/9043/ed837.png)


