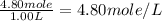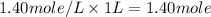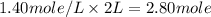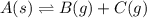Chemistry, 31.07.2019 21:20 adaakbulut9
A4.80 mol sample of solid a was placed in a sealed 1.00 l container and allowed to decompose into gaseous b and c. the concentration of b steadily increased until it reaches 1.40 m, where it remained constant. a(s)↽−−⇀b(g)+c(g) then, the container volume was doubled and equilibrium was re‑established. how many moles of a remain

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 14:10
Aconcentrated solution of ammonia is 14.8m and has a density of 0.899g/l. what is the concentration of ammonia in this solution in weight percent (%w/w)?
Answers: 1

Chemistry, 23.06.2019 07:00
Ajar contains a certain substance. which observation would show that the substance must be either a solid or a liquid?
Answers: 1

Chemistry, 23.06.2019 08:30
Explain how to convert from one unit to another in the metric system.
Answers: 3

Chemistry, 23.06.2019 09:00
How many moles of potassium hydroxide are needed to completely react with 2.94 moles of aluminum sulfate according to the following equation:
Answers: 2
You know the right answer?
A4.80 mol sample of solid a was placed in a sealed 1.00 l container and allowed to decompose into ga...
Questions




English, 24.09.2019 22:10

History, 24.09.2019 22:10

Biology, 24.09.2019 22:10










Mathematics, 24.09.2019 22:10