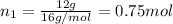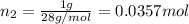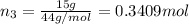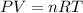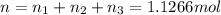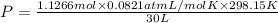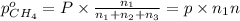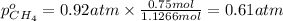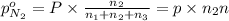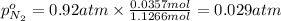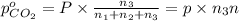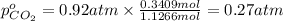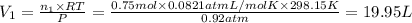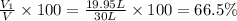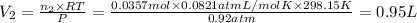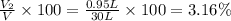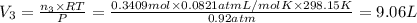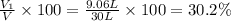Chemistry, 24.07.2019 16:10 jacksonhoyt8049
A30-liter volume of gas at 25°c contains 12 g of methane, 1 g of nitrogen, and 15 g of carbon dioxide. calculate (a) the moles of each gas present, (b) the partial pressure exerted by each gas, (c) the total pressure exerted by the mixture, and (d) the percentage by volume of each gas in the mixture. you may assume ideal gas behavior

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 16:30
What is the force of attraction between the particles in a salt crystal
Answers: 2

Chemistry, 21.06.2019 18:00
When the following equation is balanced using the smallest possible integers, what is the coefficent of oxygen gas? c7h16o(g) + o2(g) → co2(g) + h2o(g) -1 -5 -8 -16 -21
Answers: 3

Chemistry, 22.06.2019 12:30
Acontrol during an experiment. might change remains constant does not exist does change
Answers: 1

Chemistry, 22.06.2019 17:30
Upon decomposition, one sample of magnesium fluoride produced 1.65 kg of magnesium and 2.56 kg of fluorine. a second sample produced 1.32 kg of magnesium. part a how much fluorine (in grams) did the second sample produce?
Answers: 2
You know the right answer?
A30-liter volume of gas at 25°c contains 12 g of methane, 1 g of nitrogen, and 15 g of carbon dioxid...
Questions

History, 09.07.2019 20:30



Mathematics, 09.07.2019 20:30



Computers and Technology, 09.07.2019 20:30

English, 09.07.2019 20:30


Health, 09.07.2019 20:30





Arts, 09.07.2019 20:30



Biology, 09.07.2019 20:30

History, 09.07.2019 20:30