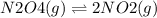Chemistry, 02.02.2020 10:44 kimlyn58p0wyn0
Which situation would cause the following equilibrium reaction to shift to the right? (1 point) n2o4 (g)two arrows stacked on top of each other. the top arrow points to the right. the bottom arrow points to the left. 2 no2
increase the pressure
add n2o4
add no2
decrease the volume

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Complete the sentence. the lower the hydrogen ion concentration, the the ph. higher lower closer to 7 closer to 0
Answers: 2

Chemistry, 21.06.2019 23:30
Two atoms interact with each other as shown by the equation. complete the equation by filling in the missing parts. 1 2 3 4 5 h he li
Answers: 2

Chemistry, 22.06.2019 12:20
Which is an example of the practical pursuit of alchemy? a. forming perfect substances. b. transforming base metals. c. developing metalworking techniques. d. linking spiritual characteristics with material substances.
Answers: 1

Chemistry, 22.06.2019 15:30
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins.co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)
Answers: 1
You know the right answer?
Which situation would cause the following equilibrium reaction to shift to the right? (1 point) n2o...
Questions

Health, 02.08.2019 23:50

Health, 02.08.2019 23:50


Health, 02.08.2019 23:50

Physics, 02.08.2019 23:50

Social Studies, 02.08.2019 23:50

Biology, 02.08.2019 23:50

Mathematics, 02.08.2019 23:50

Social Studies, 02.08.2019 23:50


Biology, 02.08.2019 23:50

Mathematics, 02.08.2019 23:50

History, 02.08.2019 23:50

Mathematics, 02.08.2019 23:50

Chemistry, 02.08.2019 23:50

Biology, 02.08.2019 23:50