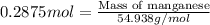Chemistry, 30.06.2019 00:00 TH3L0N3W0LF
Considering the limiting reactant, what is the mass of manganese produced from 25.0 g of manganese(iv) oxide (86.94 g/mol) and 25.0 g of aluminum metal? 3 mno2(l) + 4 al(l) 3 mn(l) + 2 al2o3(s)

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:00
Given sno2 + 2h2 - sn + 2h20 tin oxide reacts with hydrogen to produce tin and water. how many moles of sno2 are needed to produce 500.0 grams of sn?
Answers: 3

Chemistry, 22.06.2019 11:50
Acompound has a molecular weight of 12.124 atomic mass units and the empirical formula c3h40. what is the molecular formula of the compound?
Answers: 3

Chemistry, 22.06.2019 19:30
Phosphorous can form an ion called phosphide, which has the formula p3−. this ion can form an ion called phosphide, which has the formula p3−. this ion properties very similar to those of pforms when a phosphorus atom loses three protonsis called a cationcontains 18 electrons
Answers: 2

You know the right answer?
Considering the limiting reactant, what is the mass of manganese produced from 25.0 g of manganese(i...
Questions

Mathematics, 12.03.2021 21:20


Health, 12.03.2021 21:20


Arts, 12.03.2021 21:20


English, 12.03.2021 21:20



Mathematics, 12.03.2021 21:20


English, 12.03.2021 21:20

Biology, 12.03.2021 21:20







Mathematics, 12.03.2021 21:20

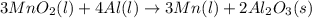
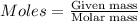 .....(1)
.....(1)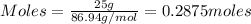
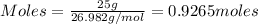
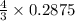 = 0.3833 moles of aluminium.
= 0.3833 moles of aluminium.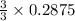 = 0.2875 moles of manganese.
= 0.2875 moles of manganese.