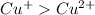Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:00
What is the main purpose of patent attorneys? defend the company against legal claims manage financial investments invent new products protect rights to new products and processes
Answers: 1

Chemistry, 22.06.2019 22:10
What is the indicator of the number of ions in solution? the amount of conductivity the amount of precipitate the amount of solute added
Answers: 1

Chemistry, 23.06.2019 04:31
How does a sample of helium at 15 degree celsius compare to a sample of helium at 215 k? a) the helium at 15 degrees celsius has a higher average kinetic energy that the sample at 215 k. b) the helium at 15 degrees celsius has lower nuclear energy that the sample at 215 k. c) the helium at 15 degrees celsius has slower- moving atoms that the sample at 215 k. d) the helium at 15 degrees celsius has smaller atoms than the sample at 215 k.
Answers: 1

Chemistry, 23.06.2019 10:50
Gene expression control that occurs during the generation of rna is a. controlled at transcription b. control before transcription c. controlled after transcription d. controlled after translation
Answers: 3
You know the right answer?
Which ion is larger? cu+ cu2+ why? because cu2+ has the filled 4s subshell. because cu2+ cannot be...
Questions

History, 31.12.2019 22:31


Biology, 31.12.2019 22:31

History, 31.12.2019 22:31


History, 31.12.2019 22:31



Mathematics, 31.12.2019 22:31

Mathematics, 31.12.2019 22:31

History, 31.12.2019 22:31

Biology, 31.12.2019 22:31

Spanish, 31.12.2019 22:31

English, 31.12.2019 22:31


Mathematics, 31.12.2019 22:31




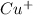 ion size is larger than
ion size is larger than 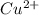 ion.
ion.