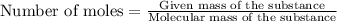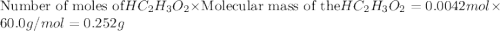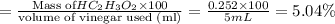The formula to determine the percent of hc2h3o2 (mass/volume) in vinegar is percent (m/v) = (grams of hc2h3o2/ volume of vinegar used (ml) ) x 100. if 5.0 ml of vinegar were used for the titration and 0.0042 moles of hc2h3o2 were required to reach the endpoint, calculate the percent of hc2h3o2 in vinegar. the molar mass of hc2h3o2 is 60.0 g / mol. question 10 options:

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 15:00
Which one of the following gases is not an important component of soil?
Answers: 2

Chemistry, 21.06.2019 17:30
Which term describes a fracture in the earth at which land stays in the same place? a. joint b. fault c. split d. hinge
Answers: 1

Chemistry, 22.06.2019 01:00
According to the tide table below what time of day will the highest tide occur?
Answers: 1

Chemistry, 22.06.2019 09:10
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2
You know the right answer?
The formula to determine the percent of hc2h3o2 (mass/volume) in vinegar is percent (m/v) = (grams o...
Questions

Mathematics, 24.08.2019 21:20


Spanish, 24.08.2019 21:20


Mathematics, 24.08.2019 21:20

Social Studies, 24.08.2019 21:20



Mathematics, 24.08.2019 21:20

Spanish, 24.08.2019 21:20


History, 24.08.2019 21:20



Biology, 24.08.2019 21:20

Computers and Technology, 24.08.2019 21:20

Health, 24.08.2019 21:20


Chemistry, 24.08.2019 21:20

Mathematics, 24.08.2019 21:20

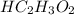 in vinegar 5.04%.
in vinegar 5.04%.