
Chemistry, 11.07.2019 07:30 emilybutler
Sulfur and oxygen form both sulfur dioxide and sulfur trioxide. when samples of these were decomposed the sulfur dioxide produced 3.49 g oxygen and 3.50 g sulfur, while the sulfur trioxide produced 6.75 g oxygen and 4.50 g sulfur. calculate the mass of oxygen per gram of sulfur for each sample and show that these results are consistent with the law of multiple proportions.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:30
If 10.g of agno3 is available, what volume of 0.25 m agno3 can be prepared
Answers: 1

Chemistry, 21.06.2019 23:50
Working with si (metric) units for each of the following commonly used measurements, indicate its symbol. liter gram milliliter kilogram meter centigram milligram centimeter kilometer second millimeter milliseconds
Answers: 1


Chemistry, 22.06.2019 18:00
Heat is the total potential energy of a substance that can be transferred. true false
Answers: 1
You know the right answer?
Sulfur and oxygen form both sulfur dioxide and sulfur trioxide. when samples of these were decompose...
Questions


Mathematics, 25.06.2019 22:00

Biology, 25.06.2019 22:00




History, 25.06.2019 22:00

Mathematics, 25.06.2019 22:00

Computers and Technology, 25.06.2019 22:00

Health, 25.06.2019 22:00


English, 25.06.2019 22:00

Health, 25.06.2019 22:00





History, 25.06.2019 22:00


Spanish, 25.06.2019 22:00

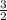 O₂
O₂


