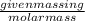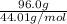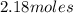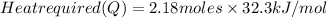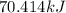Chemistry, 11.07.2019 19:00 kyrabrown33
Calculate the amount of heat required to completely sublime 96.0 g of solid dry ice (co2) at its sublimation temperature. the heat of sublimation for carbon dioxide is 32.3 kj/mol. calculate the amount of heat required to completely sublime 96.0 g of solid dry ice (co2) at its sublimation temperature. the heat of sublimation for carbon dioxide is 32.3 kj/mol.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:30
How many grams of co(g) are there in 74.5 ml of the gas at 0.933 atm and 30o c?
Answers: 1


Chemistry, 22.06.2019 05:50
In an exothermic reaction the bonding energy of the product is: less than the reactants same as the reactants greater than the reactants dependent upon the presence of a catalyst
Answers: 1

You know the right answer?
Calculate the amount of heat required to completely sublime 96.0 g of solid dry ice (co2) at its sub...
Questions


Mathematics, 06.03.2021 04:40






History, 06.03.2021 04:50


Advanced Placement (AP), 06.03.2021 04:50



Geography, 06.03.2021 04:50

Arts, 06.03.2021 04:50




Mathematics, 06.03.2021 04:50


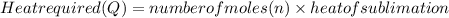 (1)
(1)