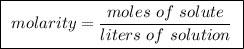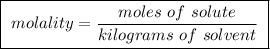Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 15:30
219 grams of iron (iii) oxide reacts with excess carbon according to the reaction equation shown below. fe2o3 + c → fe + co2 after a scientist performs the chemical reaction they find the actual yield of iron to be 57.4 grams. calculate the percent yield of this chemical reaction.
Answers: 1

Chemistry, 21.06.2019 20:30
What problem would a person have if the nucleic acid in one of his or her cells were damaged?
Answers: 2

Chemistry, 22.06.2019 06:30
Suppose a lab group reports a ppercent yield of sand of 105. is it really possible to collect more sand than was originally represented? what is the possible explanation for the extra product?
Answers: 2

Chemistry, 22.06.2019 10:30
Balance and in which category does it fit in? single or double displacement or synthesis or decomposition? (a) k2 o → k + o2 (b) na + i2 → nai (c) cu(no3 )2 + naoh → cu(oh)2 + nano3 (d) kclo3 → kcl + o2 (e) ca(no3 )2 + hbr → cabr2 + hno3 (f) sn(oh)2 → sno + h2 o (g) p4 + n2 o → p4 o6 + n2 (h) fe + al2 (so4 )3 → feso4 + al (i) alcl3 + na2 co3 → al2 (co3 )3 + nacl (j) c3 h6 + o2 → co2 + h2 o
Answers: 1
You know the right answer?
Question 4 to make a 0.500 m solution, one could take 0.500 moles of solute and add 1.00 l of solven...
Questions

English, 18.03.2021 19:10

Mathematics, 18.03.2021 19:10

Mathematics, 18.03.2021 19:10


Chemistry, 18.03.2021 19:10


Mathematics, 18.03.2021 19:10


Social Studies, 18.03.2021 19:10

History, 18.03.2021 19:10




History, 18.03.2021 19:10

English, 18.03.2021 19:10

Mathematics, 18.03.2021 19:10

Mathematics, 18.03.2021 19:10

Mathematics, 18.03.2021 19:10