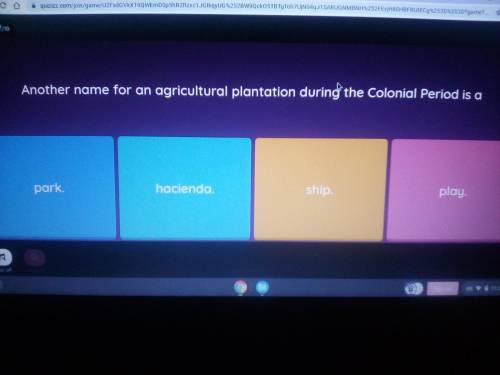
Hury hellpp me
...

Answers: 1


Another question on Social Studies


Social Studies, 22.06.2019 10:00
The history of ethical regulations in human subjects research began with the: a. declaration of helsinkib. nuremburg codec. common ruled. belmont report
Answers: 1

Social Studies, 22.06.2019 10:40
Awaiver of the requirement for documentation of informed consent may be granted when: the investigator has no convenient place to store signed consent forms separate from the research datathe only record linking the subject and the research is the consent document and the principal risk is a breach of confidentiality.potential subjects might find some of the research questions embarrassing, personal, or intrusivethe subjects are literate in their own language; however, they do not read, write, or speak english
Answers: 3

Social Studies, 22.06.2019 10:40
The specific department of health and human services (hhs) regulations that apply to research with children are known as: subpart b: additional protections for children involved as subjects in researchsubpart a: the common rulesubpart d: additional protections for children involved as subjects in researchsubpart c: protections for children in research
Answers: 1
You know the right answer?
Questions




English, 08.04.2021 09:00





History, 08.04.2021 09:00


Medicine, 08.04.2021 09:00

Social Studies, 08.04.2021 09:00





Mathematics, 08.04.2021 09:00


Mathematics, 08.04.2021 09:00

Mathematics, 08.04.2021 09:00