Physics, 31.07.2019 03:30 reneewilliams20
The specific heat of iron is 0.11 cal/goc. a cafeteria fork made of iron has a mass of 20 grams. how much heat energy is needed to raise the temperature of this form from 25oc to 75oc? 0.044 calories 0.275 calories 110 calories 9090.9 calories

Answers: 1


Another question on Physics

Physics, 22.06.2019 00:30
Ablock of steel has a density of 0.29lbs per cubic inch. if the block has dimensions of 1 inch by 1 inch by 3 inches, what is its weight? a. 0.29lbs b. 0.58lbs c. 0.87lbs d. 1lbs
Answers: 1

Physics, 22.06.2019 17:50
Convection occurs when thermal energy is transferred by the movement of
Answers: 1

Physics, 22.06.2019 18:30
Asled is being held at rest on a slope that makes an angle θ with the horizontal. after the sled is released, it slidesa distance d1 down the slope and then covers the distance d2 alongthe horizontal terrain before stopping. find the coefficient ofkinetic friction μk between the sled and the ground, assumingthat it is constant throughout the trip. find the coefficient of kinetic frictionμk. express your answer in terms of someor all of the variables d1, d2, and θ.
Answers: 1

Physics, 22.06.2019 18:30
4. now look at the green lines you created by connecting the three boiling point data points and the three melting point data points. for each of these lines, describe any trends you see. 5. locate the elements on your periodic table that you circled in green on your graph. what term or description would you use to identify these elements with respect to the periodic table? 7. using the room temperature line (orange line) and your periodic table, make lists that identify the state of matter (gas, liquid, or solid) in which each element you plotted exists at room temperature. explain your answers.
Answers: 2
You know the right answer?
The specific heat of iron is 0.11 cal/goc. a cafeteria fork made of iron has a mass of 20 grams. how...
Questions









Physics, 13.03.2020 19:48

History, 13.03.2020 19:48









Computers and Technology, 13.03.2020 19:48

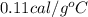 .
. 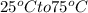
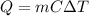
 of a mass m having specific heat C.
of a mass m having specific heat C.