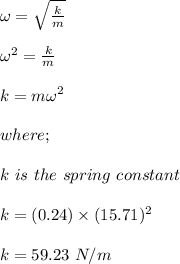Answers: 2


Another question on Physics

Physics, 22.06.2019 03:50
Three different objects, all with different masses, are initially at rest at the bottom of a set of steps. each step is of uniform height . the mass of each object is a multiple of the base mass : object 1 has mass 4..00m , object 2 has mass 1..96m , and object 3 has mass . when the objects are at the bottom of the steps, define the total gravitational potential energy of the three-object system to be zero. if the objects are then relocated as shown, what is the new total potential energy of the system? each answer requires the numerical coefficient to an algebraic expression. each algebraic expression is given using some combination of the variables , , and , where is the acceleration due to gravity. enter only the numerical coefficient. (example: if the answer is 1..23mgd , just enter 1.23)
Answers: 3

Physics, 22.06.2019 08:30
A17,250 kg rocket is pushed with a thrust of 6,450,284 n. what is the acceleration of the rocket?
Answers: 1

Physics, 22.06.2019 11:00
The dot diagram represents the motion of the motorcycle as it moves from left to right
Answers: 3

Physics, 22.06.2019 11:30
Achlorine atom has 17 protons and 18 neutrons. what is its mass number? what is its atomic number?
Answers: 2
You know the right answer?
A mass of 240 grams oscillates on a horizontal frictionless surface at a frequency of 2.5 Hz and wit...
Questions


English, 17.04.2020 21:30







Mathematics, 17.04.2020 21:30

Biology, 17.04.2020 21:30


History, 17.04.2020 21:30



English, 17.04.2020 21:30




English, 17.04.2020 21:30