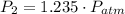To determine the pressure in a fluid at a given depth with the air-filled cartesian diver, we can employ Boyle's law, which states that the pressure in an ideal gas (held at constant temperature) is inversely proportional to its volume. At a fluid's surface, the pressure of the fluid is the same as the pressure of the atmosphere just above it, which we'll denote as LaTeX: P_{atm}P a t m. If the volume of air, which can be treated as an ideal gas here, in the cartesian diver decreases by 19% as it is lowered to a specific depth in the fluid, the pressure of the fluid at this depth, in terms of atmospheric pressure, is

Answers: 1


Another question on Physics

Physics, 21.06.2019 20:00
When using two-wire cable to feed a 240-v appliance that does not require a neutral wire, you should a. mark both ends of the white wire green. b. use the cable as is. c. use only the black wire. d. mark both ends and any exposed area of the white wire black.
Answers: 1


Physics, 22.06.2019 06:30
From 0 to 5 seconds john pushed a box 5 meters. from 5 to 10 seconds, paul pushed the same box another 5 meters. who did more work? a. john b. paul c. john and paul did the same amount of work.
Answers: 1

Physics, 22.06.2019 06:30
=force × distance a. work b. velocity c. pressure d. momentum
Answers: 1
You know the right answer?
To determine the pressure in a fluid at a given depth with the air-filled cartesian diver, we can em...
Questions















History, 22.06.2019 00:00





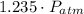 .
.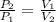 (1)
(1)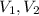 - Initial and final volume.
- Initial and final volume.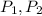 - Initial and final pressure.
- Initial and final pressure.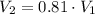 , then the pressure ratio is:
, then the pressure ratio is: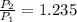
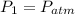 , then the final pressure of the gas is:
, then the final pressure of the gas is: