Ethan made a diagram to compare examples of the first
and second laws of thermodynamics.
...

Ethan made a diagram to compare examples of the first
and second laws of thermodynamics.
What belongs in the areas marked X and Y?
First Law of
Thermodynamics
Second Law of
Thermodynamics
X: The internal energy of a piston system increases as
heat is added and rises to a cool area
Y: As thermal energy is added to a container, some of
the energy is lost to the environment
X: As thermal energy is added to a container, some of
the energy is lost to the environment
Y: The internal energy of a piston system increases as
heat is added and rises to a cool area
X: A cold spoon placed in hot liquid gets warmer
Y: Thermal energy is converted to light energy
X: Thermal energy is converted to light energy
Y: A cold moon placed in hot liquid gets warmer

Answers: 1


Another question on Physics

Physics, 22.06.2019 12:20
The diagram shows four locations in the electric field of a positive point charge m. at which location is the electric potential the greatest.
Answers: 2

Physics, 22.06.2019 14:30
Increasing the pressure on one spot of a fluid will increase the pressure everywhere else in the fluid a. pascals principal b. archimedes' principal c. bernoulli's principal
Answers: 1

Physics, 22.06.2019 16:30
Acalorimeter uses the principle that thermal energy flows from hotter material to colder material until both reach the same
Answers: 1

Physics, 22.06.2019 18:30
Abaseball is thrown from the outfield toward the catcher. when the ball reaches its highest point, which statement is true? (a)its velocity is not zero, but its acceleration is zero. (b) its velocity and its acceleration are both zero. (c) its velocity is perpendicular to its acceleration. (d) its acceleration depends on the angle at which the ball was thrown. (e) none of the above statements are true.
Answers: 1
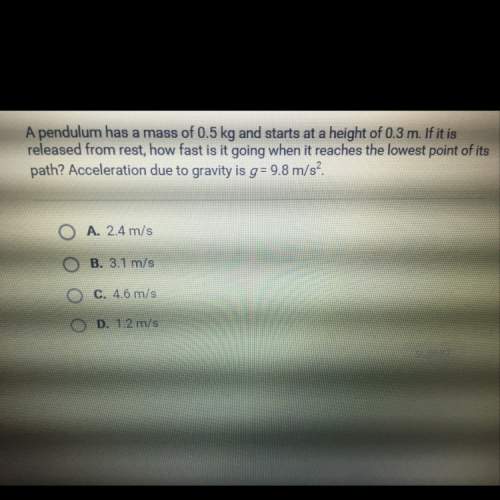
You know the right answer?
Questions


Mathematics, 13.10.2019 15:10


Advanced Placement (AP), 13.10.2019 15:10

Mathematics, 13.10.2019 15:10

History, 13.10.2019 15:10

History, 13.10.2019 15:10

Mathematics, 13.10.2019 15:10


Mathematics, 13.10.2019 15:10



Social Studies, 13.10.2019 15:10




Social Studies, 13.10.2019 15:10



Chemistry, 13.10.2019 15:10