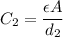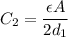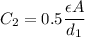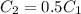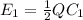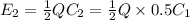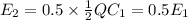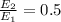Answers: 1


Another question on Physics

Physics, 22.06.2019 05:50
Acylinder with a movable piston contains 11.7 moles of a monatomic ideal gas at a pressure of 1.32×10^5 pa. the gas is initially at a temperature of 300 k. an electric heater adds 43200 j of energy into the gas while the piston moves in such a way that the pressure remains constant. cp=20.79 j k^−1 mol^−1 for a monatomic ideal gas, and that the number of gas molecules is equal to avogadro's number (6.022×10^23) times the number of moles of the gas. (a) what is the temperature of the gas after the energy is added? (b) what is the change in volume of the gas? (c) how much work is done by the gas during this process?
Answers: 3

Physics, 22.06.2019 11:50
The scalar triple product computes the magnitude m of the moment of a force vector f about a specified line. it is m = (r × f) · n, where r is the position vector from the line to the point of application of the force and n is a unit vector in the direction of the line. use matlab to compute the magnitude m for the case where f = [12, −5, 4] n, r = [−3, 5, 2] m, and n = [6, 5, −7].
Answers: 3

Physics, 22.06.2019 17:40
The first-order reaction a→2b+c has rate constant 0.538 s−1. if the initial concentration of a is 0.867 mol l−1, what is the half-life of the reaction in seconds? remember to use correct significant figures in your answer (round your answer to the nearest hundredth). do not include units in your response.
Answers: 3

Physics, 22.06.2019 18:00
Which statement is the best definition of an atom? a) anything that has mass and occupies space. b) the smallest particle that has the properties of an element. c) a substance that cannot be broken down chemically into simpler substances. eliminate d) the smallest unit of a substance that exhibits all of the properties characteristic of that substance.
Answers: 2
You know the right answer?
A parallel-plate capacitor is charged and then is disconnected from the battery. By what factor does...
Questions



Health, 16.09.2019 02:00


English, 16.09.2019 02:00






History, 16.09.2019 02:00

English, 16.09.2019 02:00

History, 16.09.2019 02:00







Biology, 16.09.2019 02:00

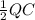 where
where 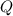 is the charge on it and
is the charge on it and 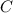 is its capacitance. The capacitance is defined, geometrically, as
is its capacitance. The capacitance is defined, geometrically, as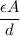
 is a constant that is determined by the material between the plates,
is a constant that is determined by the material between the plates, 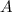 is the area of the plates and
is the area of the plates and 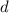 is the distance between them. It is then that the capacitance is linearly inversely proportional to the separation distance; as the distance increases, the capacitance reduces.
is the distance between them. It is then that the capacitance is linearly inversely proportional to the separation distance; as the distance increases, the capacitance reduces.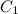 at initial separation,
at initial separation, 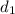
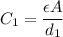
 . Then the capacitance becomes
. Then the capacitance becomes