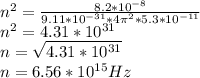In the bohr model of the hydrogen atom, an electron({rm mass}; m=9.1; times 10^{ - 31; }{rm kg}) orbits a proton at a distance of 5.3 times 10^{ - 11; }{rm m}. the proton pulls on the electron with an electricforce of 8.2 times 10^{ - 8}; {rm n}. how many revolutions per second does theelectron make?

Answers: 2


Another question on Physics

Physics, 22.06.2019 15:20
Arigid tank is divided into two equal parts by a partition. one part of the tank contains 3 kg of compressed liquid water at 400 kpa and 60°c while the other part is evacuated. the partition is now removed, and the water expands to fill the entire tank. determine the entropy change of water during this process, if the final pressure in the tank is 40 kpa. use steam tables.
Answers: 3

Physics, 22.06.2019 17:50
Which of the following best describes internal energy? a. the difference between the kinetic and potential energies of the particles in a system b. the sum of the kinetic and potential energies of the particles in a system c. the sum of the kinetic and thermal energies of the particles in a system d. the difference between the kinetic and thermal energies of the particles in a system
Answers: 2

Physics, 22.06.2019 19:30
Water is siphoned from a large tank and discharges into the atmosphere through a 50-mm diameter tube. the end of the tube is b = 2.6 m below the tank bottom which is a = 6.7 m deep, and viscous effects are negligible. determine the maximum height h over which the water can be siphoned without cavitation occurring. atmospheric pressure is 101.4 kpa, and the water vapor pressure is 1.79 kpa (absolute). report your answer in meters to two decimal places.
Answers: 1

Physics, 22.06.2019 21:30
Aperson touches a large chunk of ice with their hand and remarks, “this is making me cold.” explain what this person is feeling. is the ice transferring “cold” to the person? is there a heat transfer occurring? explain.
Answers: 1
You know the right answer?
In the bohr model of the hydrogen atom, an electron({rm mass}; m=9.1; times 10^{ - 31; }{rm kg}) or...
Questions

Computers and Technology, 27.11.2021 01:00



Chemistry, 27.11.2021 01:00

Computers and Technology, 27.11.2021 01:00




Chemistry, 27.11.2021 01:00

Health, 27.11.2021 01:00

Mathematics, 27.11.2021 01:00

Computers and Technology, 27.11.2021 01:00


Business, 27.11.2021 01:00


Mathematics, 27.11.2021 01:00

Biology, 27.11.2021 01:00



Computers and Technology, 27.11.2021 01:00