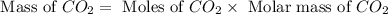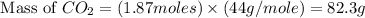Problem page gaseous ethane will react with gaseous oxygen to produce gaseous carbon dioxide and gaseous water . suppose 28. g of ethane is mixed with 190. g of oxygen. calculate the maximum mass of carbon dioxide that could be produced by the chemical reaction. round your answer to significant digits. clears your work. undoes your last action. provides information about entering answers. g

Answers: 1


Another question on Physics


Physics, 22.06.2019 06:30
From 0 to 5 seconds john pushed a box 5 meters. from 5 to 10 seconds, paul pushed the same box another 5 meters. who did more work? a. john b. paul c. john and paul did the same amount of work.
Answers: 1

Physics, 22.06.2019 11:30
1. a camcorder has a power rating of 19 watts. if the output voltage from its battery is 7 volts, what current does it use?answer units 2. a 1.5m wire carries a 6 a current when a potential difference of 57 v is applied. what is the resistance of the wire? your
Answers: 2

Physics, 22.06.2019 22:00
Aparticle of mass m is placed in a one-dimensional box of length l. the box is so small that the particle’s motion is relativistic, so that e p2/2m is not valid. (a) derive an expression for the energy levels of the particle using the relativistic energy–momentum relation and the quantization of momentum that derives from connement. (b) if the particle is an electron in a box of length l 1.00 1012 m, nd its lowest possible kinetic energy. by what percent is the nonrelativistic formula for the energy in error?
Answers: 1
You know the right answer?
Problem page gaseous ethane will react with gaseous oxygen to produce gaseous carbon dioxide and gas...
Questions

English, 23.07.2019 17:00

English, 23.07.2019 17:00


English, 23.07.2019 17:00







Social Studies, 23.07.2019 17:00


Social Studies, 23.07.2019 17:00

Biology, 23.07.2019 17:00

History, 23.07.2019 17:10




Arts, 23.07.2019 17:10

Arts, 23.07.2019 17:10

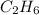 = 28 g
= 28 g = 190 g
= 190 g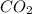 = 44 g/mole
= 44 g/mole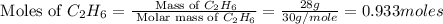

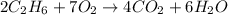
 moles of
moles of 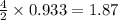 moles of
moles of